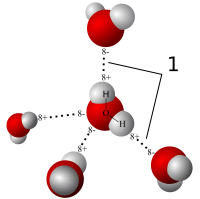
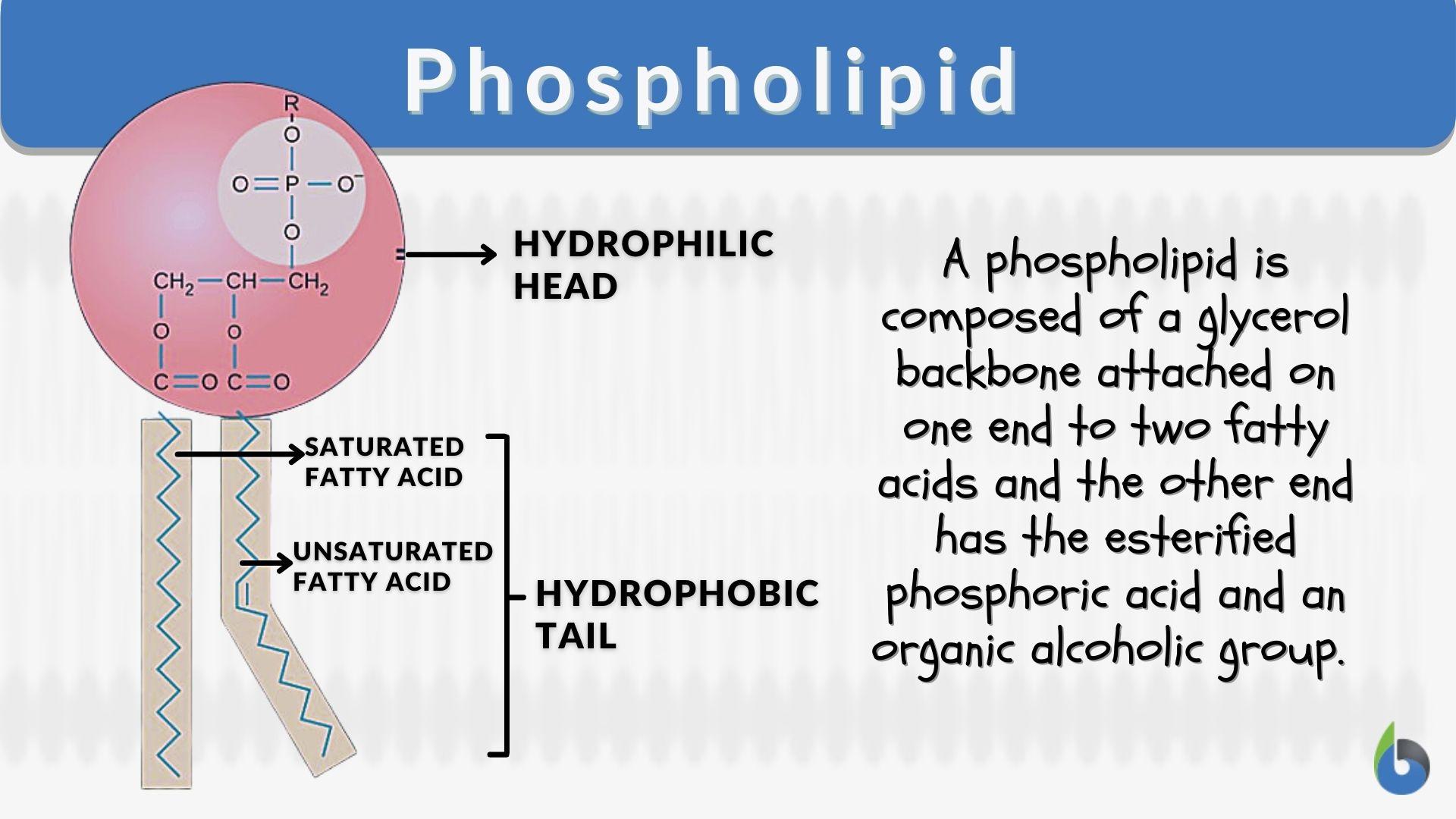
Since the head of the phospholipid contains both glycerol (non-polar) and phosphate (polar) group, why is the head considered polar overall? - Quora
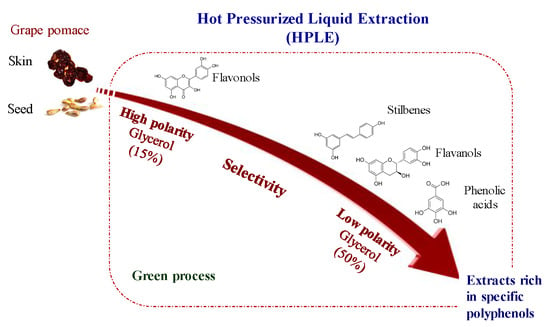
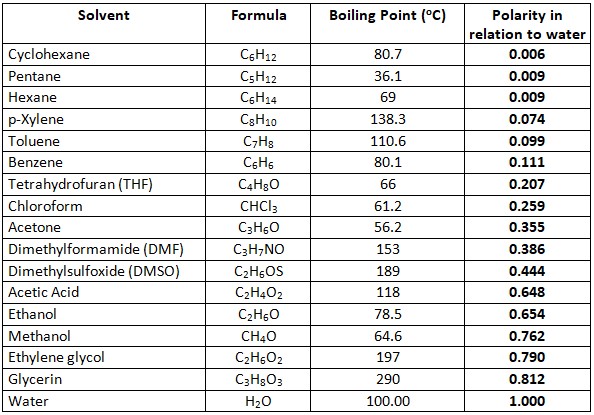
Biomolecules | Free Full-Text | Glycerol as Alternative Co-Solvent for Water Extraction of Polyphenols from Carménère Pomace: Hot Pressurized Liquid Extraction and Computational Chemistry Calculations

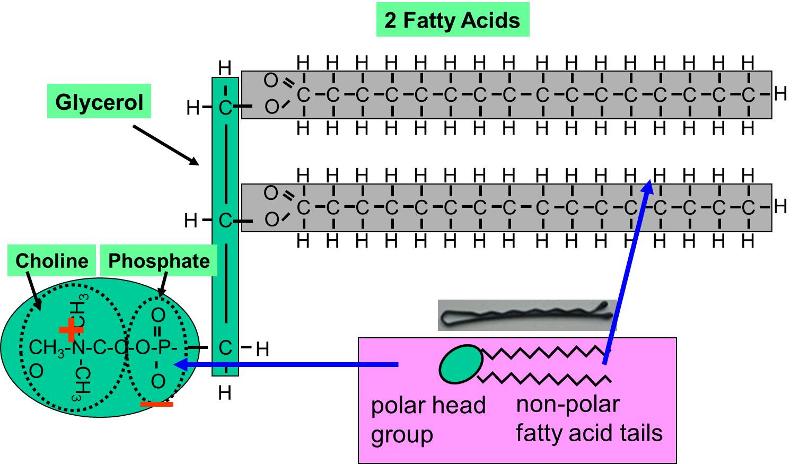
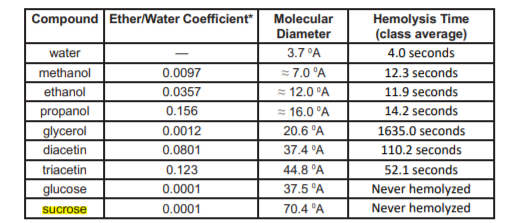
SOLVED: Phosphate 44; H;C-N-CHz-CHz-0 P-0-CHz CH; 0 H H Choline KC-0-cc-C-C H H C-C-C- H HH H H H A 0 H h h hhHhhhhhhhw #c-0-c-C-c-C-C-C-C- C-C C-C- C-C-C-C-C-C Polar Glycerol backbone

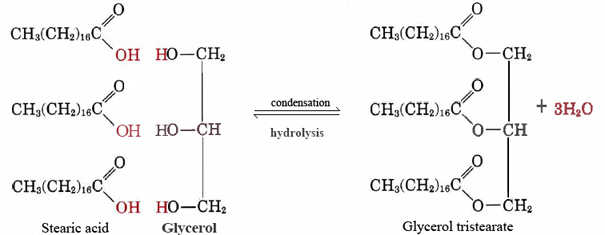
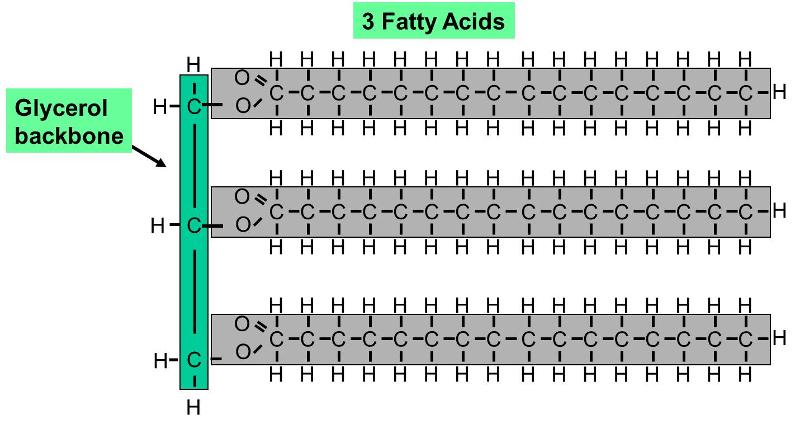
The triglyceride and the glycerol differ with respect to polarity and H-bonding. Discuss these two differences in terms of their chemical structures. | Homework.Study.com
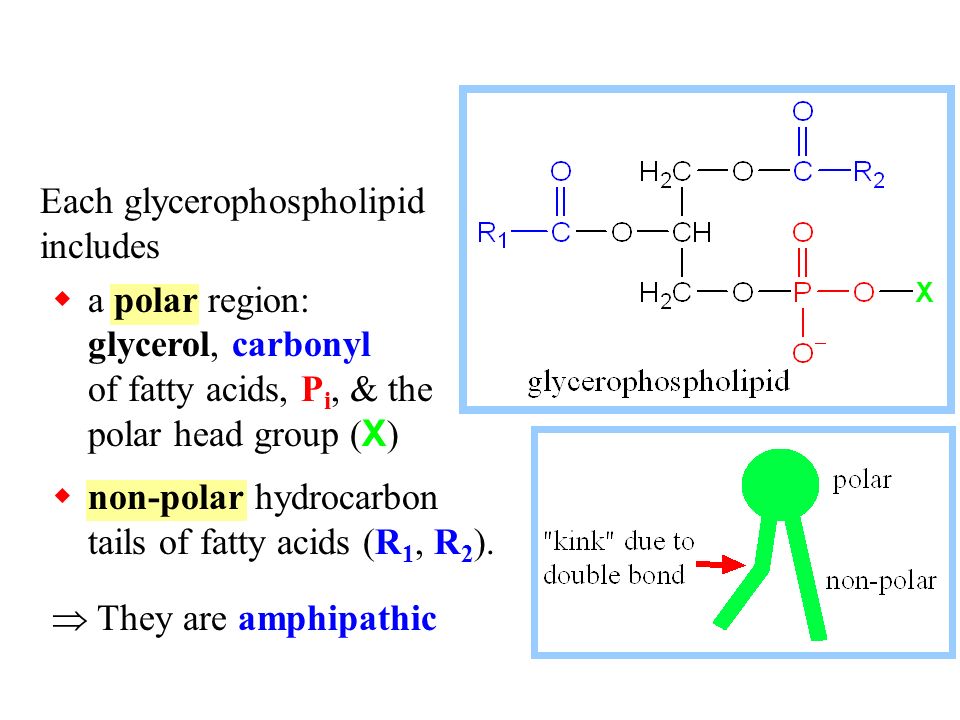
Lipids. Lipids are non-polar (hydrophobic) compounds, soluble in organic solvents. 1. Simple lipids: esters of FA with alcohols Fats: alcohol = glycerol. - ppt download
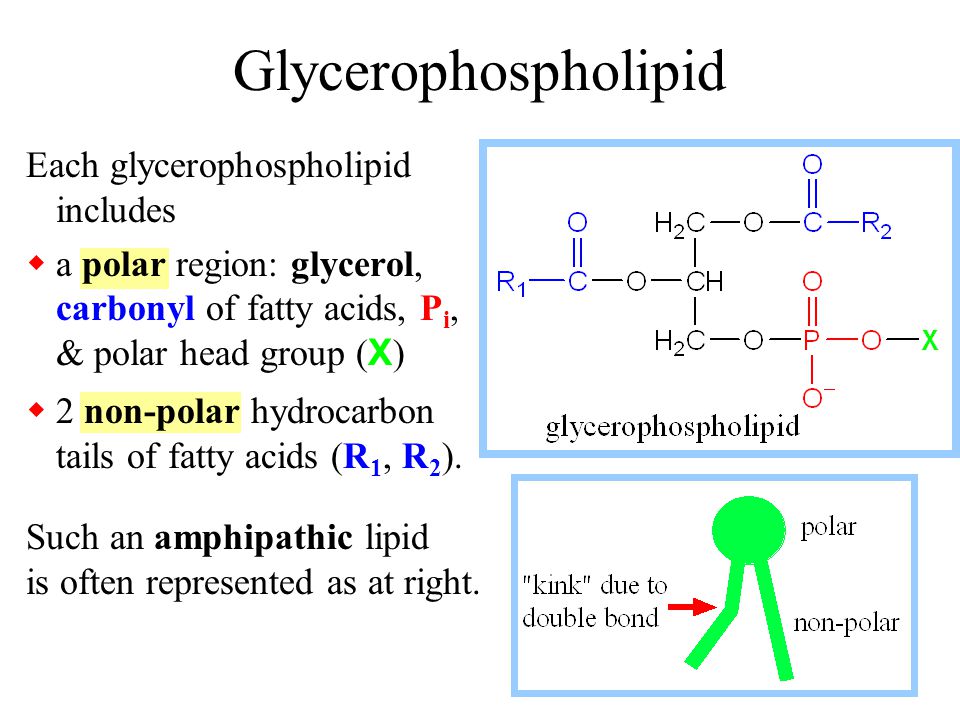
Lipids & Membranes. Lipids Lipids are non-polar (hydrophobic) compounds, soluble in organic solvents. Most membrane lipids are amphipathic, having a non-polar. - ppt download