No7 Laboratories Line Correcting Booster Serum, Targeted, Results-Driven Formula, Suitable for Sensitive Skin, 0.84 Ounce - Walmart.com

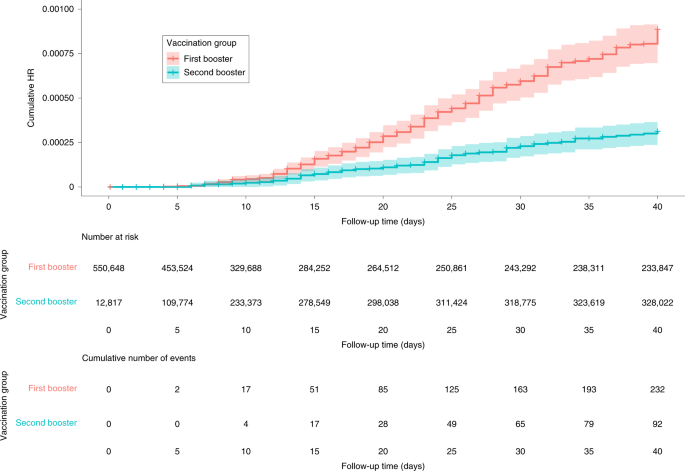
Efficacy, safety, and immunogenicity of a booster regimen of Ad26.COV2.S vaccine against COVID-19 (ENSEMBLE2): results of a randomised, double-blind, placebo-controlled, phase 3 trial - The Lancet Infectious Diseases

Effectiveness of homologous and heterologous booster doses for an inactivated SARS-CoV-2 vaccine: a large-scale prospective cohort study - The Lancet Global Health

COVID-19 vaccine waning and effectiveness and side-effects of boosters: a prospective community study from the ZOE COVID Study - The Lancet Infectious Diseases

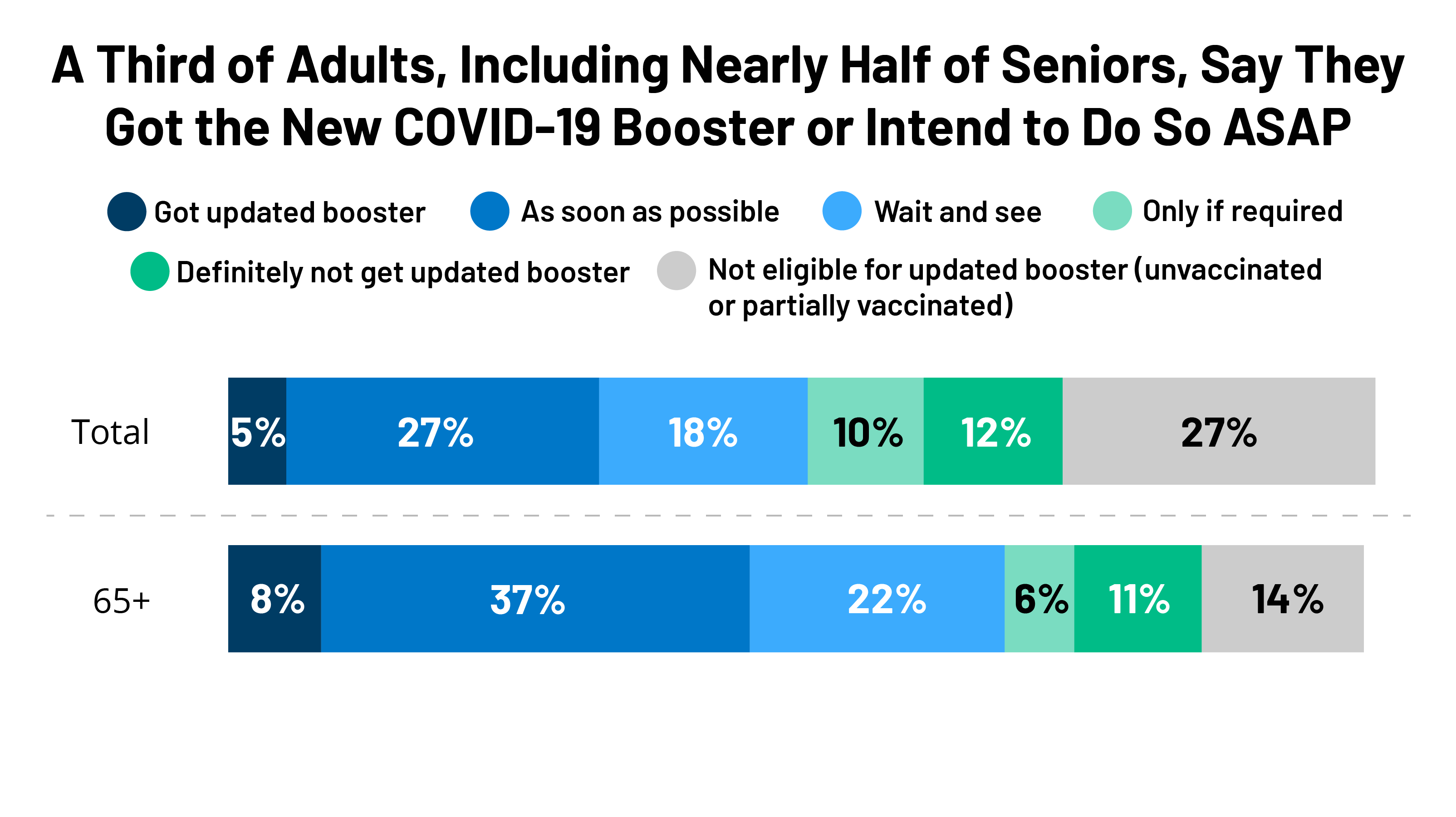
Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...

No Increased Stroke Risk Linked to Pfizer's Covid Boosters, Federal Officials Say - The New York Times

Current evidence on efficacy of COVID‐19 booster dose vaccination against the Omicron variant: A systematic review - Chenchula - 2022 - Journal of Medical Virology - Wiley Online Library

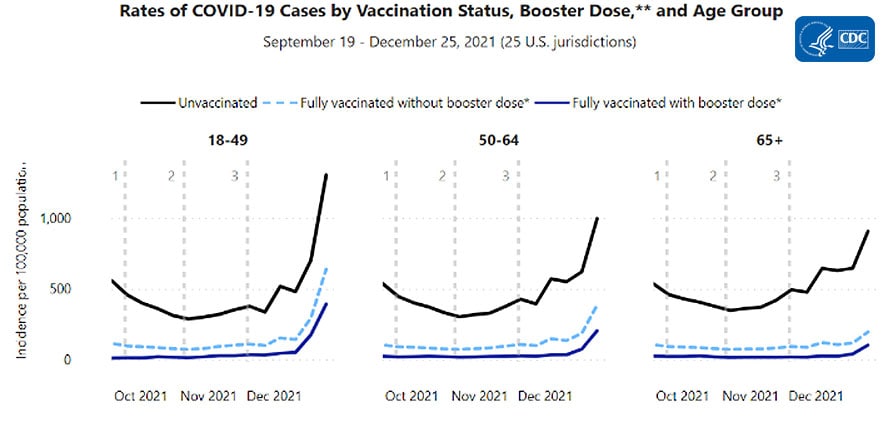
COVID-19 Incidence and Death Rates Among Unvaccinated and Fully Vaccinated Adults with and Without Booster Doses During Periods of Delta and Omicron Variant Emergence — 25 U.S. Jurisdictions, April 4–December 25, 2021 | MMWR

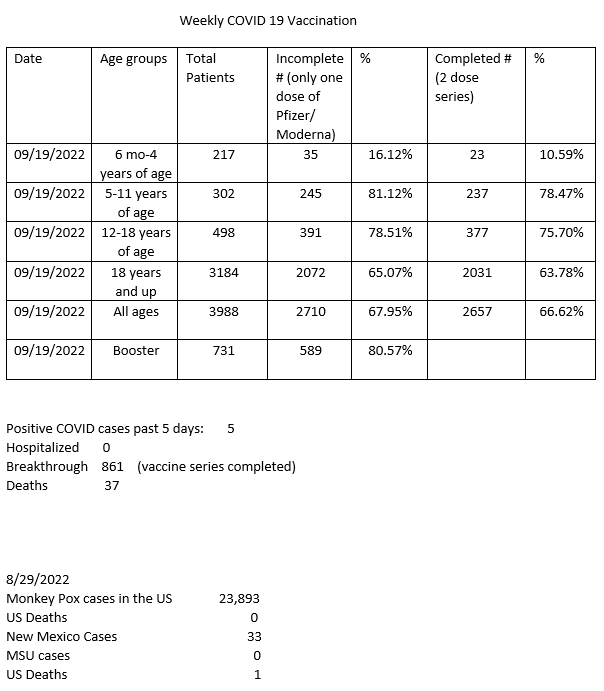
Effectiveness of emergency declarations, booster vaccinations, and vaccine passports | COVID-19 AI and Simulation Project

Antibody persistence 2 and 3 years after booster vaccination of adolescents with recombinant acellular pertussis monovalent aPgen or combined TdaPgen vaccines - eClinicalMedicine

Effectiveness of Bivalent mRNA Vaccines in Preventing Symptomatic SARS-CoV-2 Infection — Increasing Community Access to Testing Program, United States, September–November 2022 | MMWR

Mixed vaccine schedules offer strong booster responses after two doses of CoronaVac vaccine CORONAVIRUSVACCINES — Target Discovery Institute
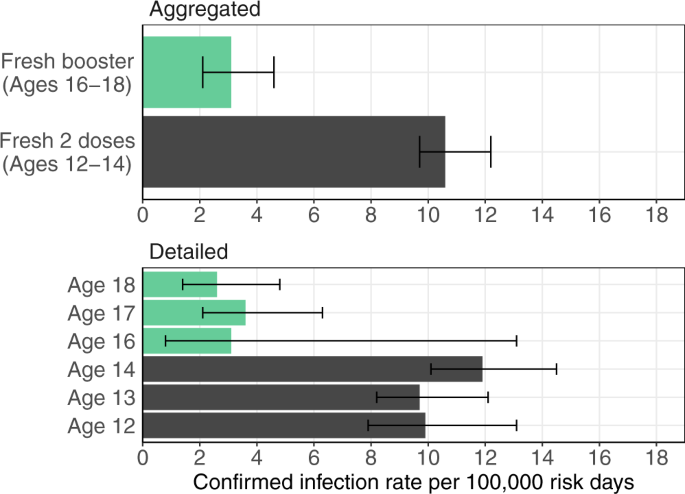
Protection following BNT162b2 booster in adolescents substantially exceeds that of a fresh 2-dose vaccine | Nature Communications

Safety and immunogenicity of seven COVID-19 vaccines as a third dose ( booster) following two doses of ChAdOx1 nCov-19 or BNT162b2 in the UK (COV-BOOST): a blinded, multicentre, randomised, controlled, phase 2 trial -

Houston Methodist's Mandate of Covid-19 Vaccine Boosters Among Health Care Workers: Setting Precedents During Unprecedented Times | Catalyst non-issue content